I started my PhD in February 2020, two weeks after the first case of Covid-19 was diagnosed in Australia. One month later, Queensland, where I am based, was in lockdown, and we spent much of the next two years under pandemic restrictions.
These were scheduled to lift in December 2021, but the state government warned that a wave of Covid-19 was coming. “Get vaccinate” was the message.
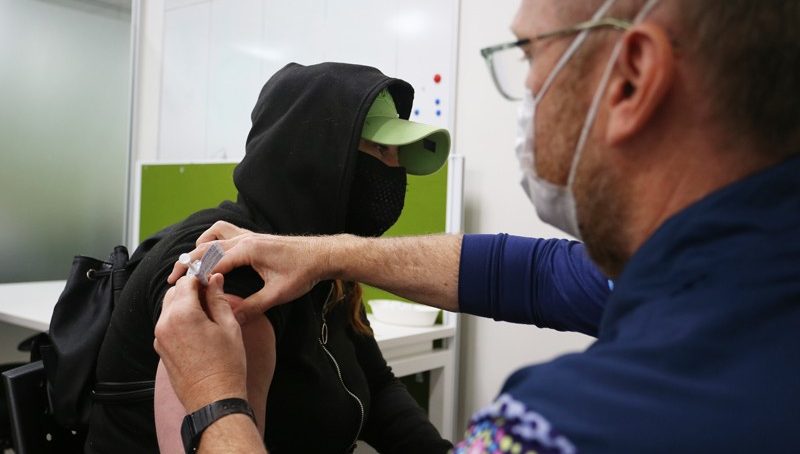
My PhD research at Griffith University in Brisbane was intended to evaluate well-being support programmes for people who are homeless. But once Covid-19 took effect this became infeasible, so I pivoted to researching vaccination strategies for people who are homeless.
To do this, I connected with Dona Hooshmand, a public-health doctor who was running mobile Covid-19 vaccination clinics for this population in southeast Queensland.
The clinics operated alongside other services for people who are homeless, including meal programmes and drop-in centres. I planned to investigate whether an incentive of ($7) would improve uptake of the second vaccination dose, and then to test whether reminder text messages would improve uptake of the third dose.
A 2014 study found that 95 per cent of people who are homeless in Australia have a mobile phone.
But the team was very small – often just me and the doctor – so I soon became involved in running the clinics. This involved designing fliers, coordinating with staff at sites, providing education about Covid-19, assisting with gaining consent, organising paperwork, doing data entry and helping with set-up. This ensured that as many people as possible could be vaccinated before the restrictions lifted in December and the wave of cases peaked.
The pandemic disrupted my research. In a clinical trial, all participants should be treated equally in every way except the intervention (in our case, the financial incentives and reminder messages).
So, I began by ensuring that all participants received vaccination reminders on the exact dates and using the exact wording specified in the trial schedule.
However, this became impractical as restrictions loosened and rates of Covid-19 infections increased. We began to run extra clinics, often at short notice as we became aware of a particular need for services. These required reminders in addition to the four planned in the schedule: people could not come to be vaccinated if they did not know we were vaccinating.
A second issue arose when participants began contacting me outside my planned schedule, seeking information about other places where they could get a vaccination if – owing to their movement out of the local area, regional travel restrictions or our scheduling – they were unable to return to our clinics.
On the one hand, ethically, participants have a right to information about new and alternative opportunities to get vaccinated. On the other hand, extra reminders with this information might have meant that these participants were more likely than those in earlier groups to present on time for their vaccinations, which was a problem, because these were key outcome measures in the trial.
The balance between participant welfare and research integrity became a major struggle for me. If you find yourself navigating a similar situation, here are four things I learnt.
At first, I turned to the literature to help me to decide how actively I should be involved in the running of the clinics. The papers I read generally agreed that a researcher should disrupt a trial process to ensure participant welfare only in a “life-threatening emergenc”’, such as an acute medical event. I began to wonder: does a pandemic in which people who are not fully vaccinated will, over time but inevitably, become sick and die count as a life-threatening emergency?
I discussed this with my supervisors as research-methodology experts, and with other PhD students, researchers, ethics advisers, the directors of the clinic sites, the clinic doctor and the participants themselves.
A common theme emerged: to prevent illness and deaths, it was crucial that I get involved at some level to ensure that people had opportunities to be vaccinated before restrictions were lifted and the Covid-19 wave peaked.
The next step was to decide exactly how, and to what extent, I would be involved. While working through this process, I learnt that I had to thoroughly understand my scope – exactly what I could and couldn’t do to maintain my impartiality as a researcher while also having a positive effect on the vaccination effort.
This is often set out when a study receives approval from an ethics board. For example, I was allowed to remind participants that their second vaccination was due, and tell them about new and alternative vaccination opportunities, but I could not give advice about whether or not they should be vaccinated.
Being clear about your scope helps you to make good decisions when put on the spot with tricky questions or requests (for instance, “Can you help me to organise my proof-of-vaccination certificate?” — absolutely. “Can you tell me if I should get the Pfizer or Moderna vaccine?” – no, but I know of someone who can).
If you are asked to act beyond your scope, you must be prepared to give a polite but firm “no”. Explain why you cannot help, remembering that people often have a limited (or even incorrect) understanding of researchers and the research process.
Then, be prepared to discuss alternatives and solutions: saying “no” is always easier if you can continue to offer help.
Flexibility became essential. This meant relaxing our rigid schedule of reminders. Rather than sending generic reminder messages to people ready to receive their vaccines (‘You are due for your second vaccine’), I sometimes waited a day or two for decisions to be made about the date, time or location of an extra clinic, and then sent a more specific reminder (‘You are due for your second vaccine, and you can see the doctor who gave your first vaccine at …’).
I also sent extra reminders to very overdue participants. These would not change the outcome that their second or third dose was not received on time, but I recognised that many of the people in question were particularly vulnerable and would benefit from extra support.
I also developed wording to respond to questions about alternative vaccination locations, which would give people the particular information they needed while being as uniform as possible.
You might find there is a need to amend your ethics approval. Or, when writing up the research, you might change a discussion point or mention amendments to the protocol as study limitations. Bear in mind that the amount of flexibility that can be tolerated depends on the nature of your research, but it is important to have at least some.
Finally, bear in mind that you cannot conduct quality research without ensuring participant welfare: each is necessary to the other.
Australia’s National Statement on Ethical Conduct in Human Research, by which I am bound, states that “respect for the participants” must not be compromised by “the aims of the research” or “the way it is carried out”.
Through collaboration, clarity of scope and flexibility, I was able to find a comfortable balance between quality research and participant welfare. In doing so, I have contributed to delivering more than 300 Covid-19 vaccinations to people who are homeless, and have run high-quality trials that will form the basis of my PhD and generate solid evidence for practice.
- A Nature opinion / Laura McCosker






